Lately, we’ve been sending off many of our herbs for pesticide testing. Even though few things are certified organic, none of our herbs are grown using pesticides to the best of our knowledge.
And that is where our policy of independent lab analysis comes in. Trust…but verify.
Laboratory testing can be harder to do than you might imagine. With our rare herbs, there often are missing reference samples, different testing methods (see about Shilajit Fulvic and Humic acids just covered, as well as trying to get the testosterone amounts in Pine Pollen for example) and more.
Such was the surprising case in pesticide testing for Shilajit.
If you’re not familiar with Shilajit, it’s a mineral pitch substance that comes out of the Himalayan mountains. It wouldn’t even need pesticides because its not technically plant material any longer, though it once was many tens of thousands of years ago.
And that’s why we were surprised to see our pesticides test come back with a failure! Note that this was only for the Powder, and not for the Resin. The Resin passed completely.
But the truth of the matter is trickier to get to. Buckle up as we dive into some chemistry and hypothesizing…
Here is the test results for our Shilajit Powder.
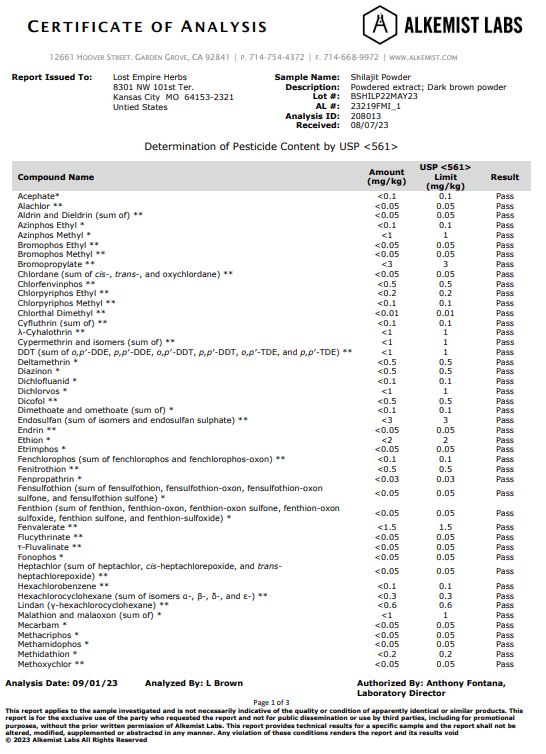
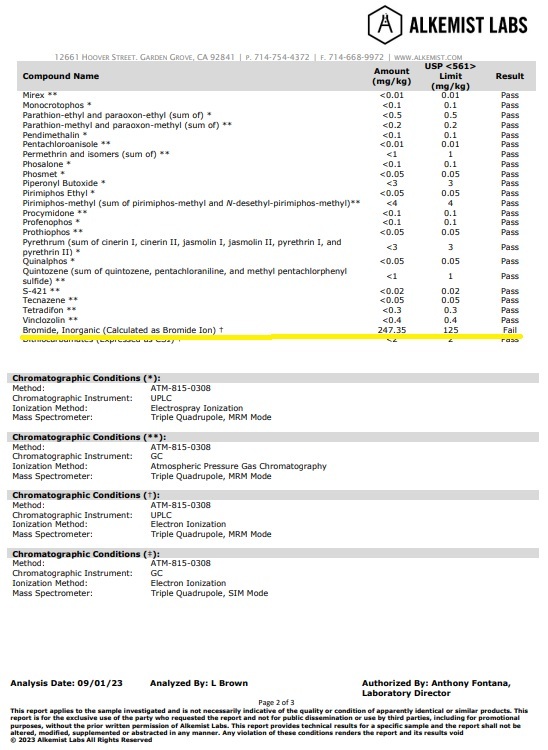
It passed every pesticide test except for one. It came back with a FAIL on “Bromide, Inorganic (Calculated as Bromide Ion)”, coming in at 247.35 mg/kg when the threshold to pass was 125 mg/kg.
Remember that we’re talking chemistry here where organic has a very specific definition that has nothing to do with use of chemical control agents. In chemistry, organic compounds are defined by the presence of carbon atoms. Inorganic compounds, on the other hand, do not contain carbon-hydrogen bonds.
Bromine is a chemical element in the periodic table which is denoted by Br. It is a halogen gas, of the same class as chlorine or iodine.
Meanwhile, bromide is the anion that forms when bromine gains an electron. It can be represented by the symbol Br–.
These bromide ions (Br-) fall under the inorganic category as they are devoid of carbon.

So why is inorganic bromide on the pesticide test?
Inorganic bromide residues typically result from fumigation with methyl bromide which is used as an antimicrobial agent and insecticide. Some research has shown that it has negative effects on the central nervous system. Even the EPA acknowledges some risks to it.

But it is the breakdown product of methyl bromide, bromide ions, that are looked for in this pesticide report.
As you know, shilajit is a mineral substance. The presence of bromide in our Shilajit could be due to natural bromide ions inherent in Shilajit, rather than contamination from a pesticide like methyl bromide.
Shilajit is a complex mineral-rich substance, formed over centuries through the gradual decomposition of plant and organic matter. Given its natural origin and high mineral content, the presence of natural bromide ions is plausible.
Even methyl bromide is sometimes produced naturally like from algae and kelp, according to that EPA report.
Overall, bromide seems to be incorporated into marine sources more commonly. Bromine is not a standard component of natural fatty acids, though brominated fatty acids can exist in some marine organisms. The same is true of phenolic lipids.
Shilajit isn’t a marine substance. But it also contains rare molecules like natural forms of C60, so again there is plausibility.
All this to say that this may be a false positive for this test.
(The Resin passing doesn’t help to shine light on this, as it simply had less than the detection level limit.)
Even if it were not a false positive and this fumigant is somehow making its way into Shilajit, the amount is very low.
The Code of Federal Regulations shows that the residue limit is 250 ppm or mg/kg for tomato, concentrated products and fig, dried fruit. There is up to 400 ppm limit for egg, dried and herb, processed and spice.
These are higher than the 247.35 mg/kg or ppm that Shilajit came back with. And you’d be sure to be eating more tomatoes, dried fruit, eggs in quantity than you would with Shilajit.
So that’s the worst case scenario. But again, our hypothesis is that these are naturally occurring bromide ions.
We will be continuing to consult with our supplier and our labs to get to the bottom of this.
- Increasing Androgen Receptor Number and Capacity - April 19, 2024
- Yang, Qi, Mitochondria and Chronic Fatigue - March 27, 2024
- Eczema: A Natural Approach to Soothing Your Skin - March 10, 2024


Leave a Reply